- English
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
Pet Rapid Test Kit
Pet Rapid Test Kit
Culture Medium
Culture Medium
Featured Products
About Us
The company has established a strict quality management system and passed the ISO9001:2008 and ISO13485:2003 international quality system certification.
It has the current international advanced automatic production equipment, and the workshop strictly follows Standard design, and reach ten thousand purification standards.
New Products
News

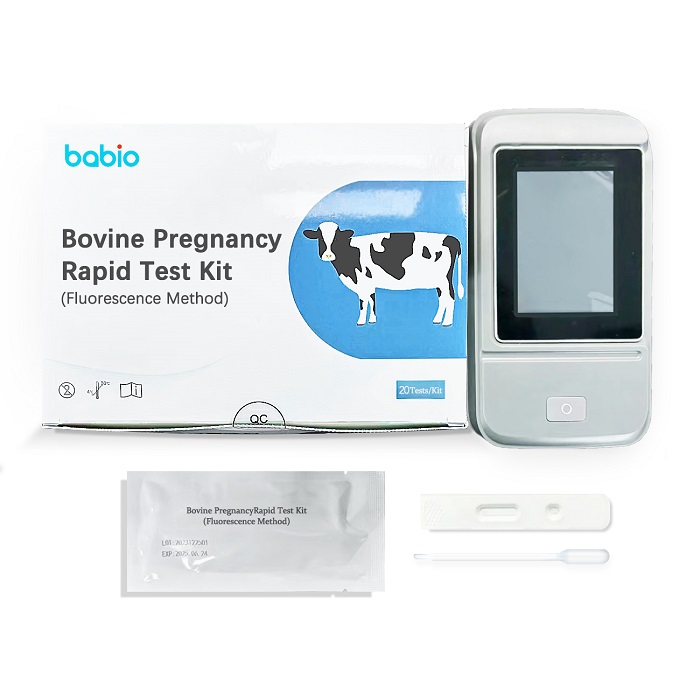
BABIO Launches Fast Bovine Pregnancy Rapid Test Kit for Modern Cattle Farming
BABIO Bovine Pregnancy Rapid Test Kit delivers fast, accurate cattle pregnancy testing with fluorescence technology for dairy and beef farms.

BABIO Salmonella Shigella Enrichment Medium Enhances Enteric Pathogen Detection
Discover BABIO Salmonella Shigella Enrichment Medium for enteric pathogen transport, food safety testing, and clinical microbiology labs.

BABIO Launches Rapid TOXO Ab Test Kit for Veterinary Screening
Rapid Toxoplasma Gondii antibody test kit for cats and dogs by BABIO. Accurate veterinary screening for fast pet infection diagnosis.

BABIO Launches High-Performance MR-VP Broth for Modern Microbiology Laboratories
Discover BABIO MR-VP Broth for accurate bacterial differentiation, food microbiology testing, and reliable Methyl Red Voges-Proskauer analysis.

BABIO Bovine Pregnancy Test Kit – Rapid Fluorescence Method
Detect cow pregnancy early with BABIO’s fluorescence rapid test kit. Accurate results in 15-20 mins. Visit https://www.babiocorp.com for details.
BABIO Salmonella Shigella Transport Medium for Labs
BABIO Salmonella Shigella Enrichment Medium ensures safe transport and accurate detection of Salmonella & Shigella in labs, hospitals, and food testing.