
- English
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
BABIO Launches High-Performance Virus Transport Kit (Non-Inactivated) for Global Laboratories
BABIO, a leading Chinese diagnostics manufacturer since 2003, has officially released its upgraded Virus Transport Kit (Non-Inactivated) to meet the rising global demand for reliable viral sample collection and transport. The solution is engineered for hospitals, research centers, reference labs, and public health facilities across Europe, the United States, Africa, and Southeast Asia.
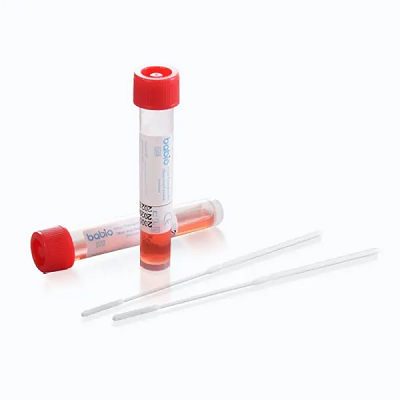
Designed for high-integrity viral preservation, the kit features a stabilized non-inactivated VTM formula based on improved Hank’s solution with multiple antibiotics to inhibit bacterial and fungal contamination. Available in 1–6 ml tubes and pack sizes from 20 to 500, the kit supports diverse testing needs including PCR, qPCR, nucleic acid extraction, and virus isolation.
Each kit includes DNase/RNase-free transport tubes with a shatter-resistant design and high-quality flocked swabs with breakpoint technology. BABIO’s strong production capacity—over 100,000 units per day—ensures fast delivery and competitive global pricing.
As a trusted supplier with CE-certified products and long-term distribution in major markets such as the U.S. and Germany, BABIO continues to expand its footprint by providing dependable viral transport solutions for pandemic preparedness, routine diagnostics, and global health programs.
For more details, please visit the official website: https://www.babiocorp.com
#VirusTransportKit #VTM #BABIO #DiagnosticReagents#LaboratoryTesting #PCRTesting #SampleCollection#GlobalHealth #MedicalSupplies #BiotechnologyChina


